 It corresponds to the charge number of the nucleus. The atomic number uniquely identifies an element chemically. It’s the 19 th most abundant element found in nature. Definitions Atomic Number of Carbon: The atomic number or the so-called proton number for any chemical element is the number of protons present in the nucleus of each atom of that chemical element. 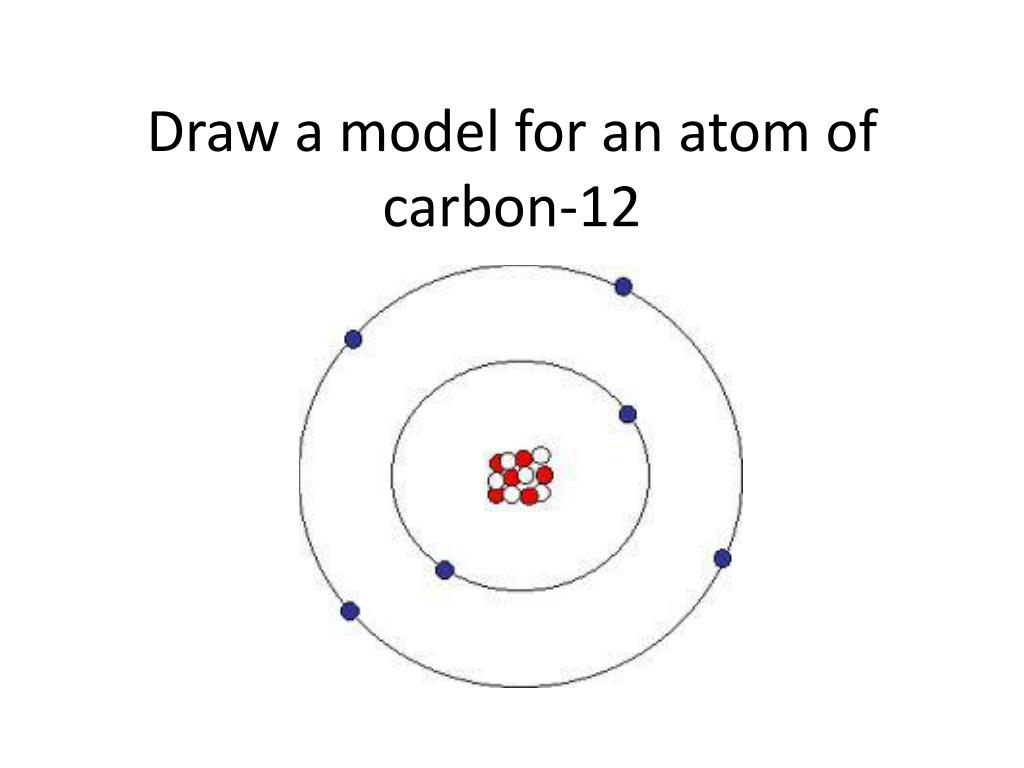
Since carbon has six protons and six neutrons, its atomic mass is 12. Lithium 3 7 Aluminum 13 14 Oxygen 8 Zinc 65 30 Cesium 55 78 Bromine 80 35 Lead 82 125 Gold 79 197 Nitrogen Calcium 20 Tin 119 50 Iodine 53 74 Silver 108 61 Neon 10 Atomic Mass # of Protons # of Neutrons # of Electrons Element Atomic # of Neutrons # of Electrons Element Atomic Mass Atomic # of Protons 40 20 20 50 69 50 53 127 53 47 47 47 10 10 10 Atomic # = # Protons = # Electrons Atomic Mass = Protons + Neutrons Neutrons = Atomic Mass – Atomic #ĥ Atomic Number 6 Protons Electrons C Carbon 12.01 A.P.E. Carbon is a chemical element with an atomic number of 6 in the periodic table of elements. Each proton and neutron is given an atomic mass of 1. 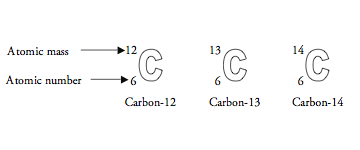
Carbon-12 is the common isotope, with carbon-13 as another stable isotope. 1 Atomic Number 6 Protons Electrons C Carbon 12.01 A.P.E.ģ Lithium 3 7 Aluminum 13 14 Oxygen 8 Zinc 65 30 Cesium 55 78 Bromine 80 35 Lead 82 125 Gold 79 197 Nitrogen Calcium 20 Tin 119 50 Iodine 53 74 Silver 108 61 Neon 10 Atomic Mass # of Protons # of Neutrons # of Electrons Element Atomic # 3 4 3 27 13 13 Do You See A Pattern? 16 8 30 35 30 133 55 35 45 35 82 207 82 79 118 79 7 14 7 Atomic # = # Protons = # Electrons Atomic Mass = Protons + Neutrons Neutrons = Atomic Mass – Atomic #Ĥ Atomic # = # Protons = # Electrons Atomic Mass = Protons + Neutrons The element is determined by the atomic number 6. 
0 Comments
Leave a Reply. |
Details
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |

 RSS Feed
RSS Feed
